BioMARC specializes in manufacturing biopharmaceutical products for non-clinical and clinical use under Biosafety Level 2 & 3 containment and Select Agent Tier 1 biosecurity conditions.
In addition to manufacturing and testing services, BioMARC offers product and process development services (both upstream and downstream), including the creation of development programs focused on implementation of cGMP regulations and scalability. BioMARC has the expertise, equipment, and facilities to take your product from preclinical through Phase I and II manufacturing development.
Process Development
Process development is a critical element of manufacturing. We believe that robust process design should be grounded in a deep understanding of underlying process behavior. Well-designed experiments and rigorous data analysis enable more efficient, high-quality decision-making.
Upstream (bulk processing) and downstream (drug materials) development studies ensure the process meets its intended use. These studies focus on generating reliable data to demonstrate the process’s reproducibility. Development is guided by critical criteria outlined in the Project Specific Plan, with decisions based on representative data. If interactions between critical process parameters and starting material properties are unclear, processes are strictly reproduced to gather meaningful data. Development includes analyzing individual steps and the full production process using pilot or engineering batches. All activities are documented to support the product’s life cycle.
Cell & Virus Banking
The concept of a two-tiered cell or virus bank, in which the Master Cell Bank (MCB or MVB) is used to generate Working Cell Banks (WCBs or WVBs), is generally accepted as the most practical approach to providing a supply of cell substrate or viruses for continued manufacture of a cell-based product.
In some cases, transient transformation of microbes, yeast or mammalian cell lines may be performed for each production lot using aliquots of appropriately tested and expanded host cell banks and plasmid/genetic material for each transformation. The transformed cell substrate is then used for preparation of the product as bulk material for further processing. This alternative transient transformation of microbial, yeast, or mammalian cells can be developed as a highly robust and reproducible process. This transformed cell substrate bank is considered the MCB, and it is used as the source of cell substrate for production. Regulatory agencies consider this alternative system adequate because the transformation of bacteria and yeast is generally a very reproducible and easily performed process, unlike the events needed for transfection of metazoan cells.
BioMARC offers cell and virus banking services as part of a larger GMP project or as an independent service. This includes evaluating the anticipated utilization rate of the bank for production, the expected intervals between generation of new banks, and the criteria for qualification of cell/virus bank(s).
Analytical Method Development
Analytical method development is important for both the early and advanced phases of the product life cycle. As such, analytical methods used in raw material analysis, in-process testing, release testing and cleaning validation/verification associated with GMP biopharmaceutical products are validated, qualified, or verified, as appropriate for the stage of the product. Development of these methods is carried out in such a way that the type and scope of the data generated will allow for sufficient evidence of the reproducibility of the process and in accordance with the industry standards of analysis (i.e., precision, accuracy, limit of detection, limit of quantification, specificity, range and robustness).
Development of a project’s methods will focus on designing studies and generating data based on critical criteria outlined in the Project Specific Plans and will include a gap analysis and remediation planning. The result of development is a method poised for use in the project life cycle.
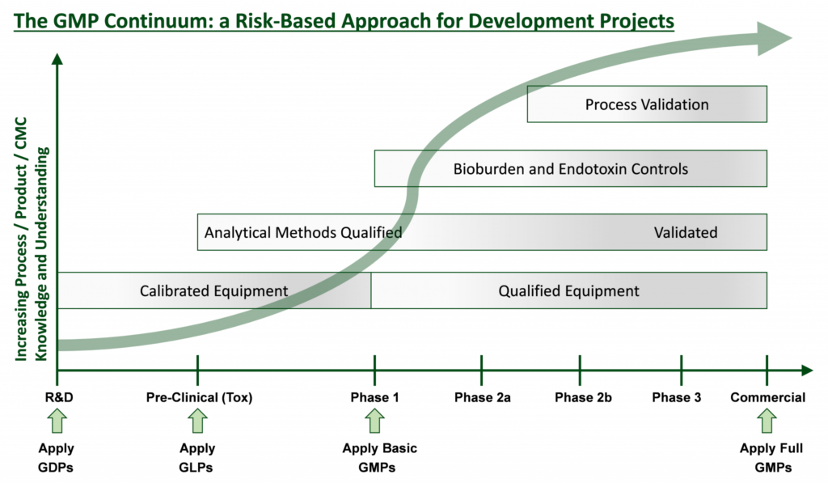
Continuum by Design
The graph below represents the appropriate regulatory framework applied at each stage of development, ensuring regulatory compliance and reasonable cost.